Intrathecal Pump Implantation
Intrathecal Pump Implantation
Introduction
Chronic pain can be devastating, especially when it does not respond to conventional treatments like oral medications, injections, or physiotherapy. Patients with cancer-related pain, severe nerve pain, or muscle spasticity often require high doses of medication, which may cause drowsiness, nausea, constipation, or even organ damage in the long term.
For such cases, advanced therapies like intrathecal pump implantation offer a breakthrough solution. By delivering medication directly to the spinal fluid, this device provides powerful pain relief with minimal side effects, allowing patients to regain independence and comfort.
What is an Intrathecal Pump?
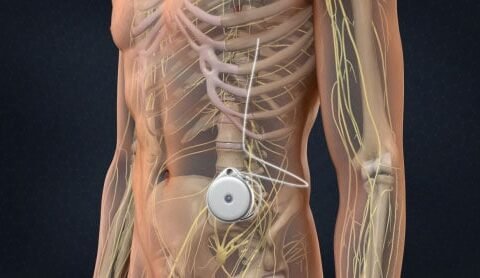
An intrathecal pump (also called a pain pump) is a small, programmable device surgically implanted under the skin of the abdomen. The pump delivers controlled amounts of medication directly into the intrathecal space, where spinal fluid surrounds the spinal cord.
Because the drug is delivered closer to the pain receptors in the spinal cord, only a fraction of the usual oral dose is needed to achieve strong relief.
Conditions Treated with Intrathecal Pump Implantation
-
Cancer-related pain not controlled by oral opioids
-
Neuropathic pain due to spinal cord injury or nerve damage
-
Failed back surgery syndrome (FBSS)
-
Complex regional pain syndrome (CRPS)
-
Severe spasticity (muscle stiffness and spasms) in conditions like multiple sclerosis or cerebral palsy
How the Procedure Works
-
Evaluation
-
The patient undergoes medical screening to ensure suitability.
-
A trial dose is given via spinal injection to test effectiveness.
-
-
Surgery
-
A small incision is made in the abdomen to place the pump.
-
A catheter is tunneled under the skin and connected to the intrathecal space near the spinal cord.
-
-
Programming
-
The pump is programmed to release medication continuously or at specific times.
-
Adjustments can be made externally by the doctor using a handheld programmer.
-
-
Maintenance
-
The pump needs periodic refilling (every 1–3 months) via a simple injection through the skin.
-
Battery life of the device is 5–10 years, after which replacement is needed.
-
Benefits of Intrathecal Pump Implantation
-
Superior pain relief with very small drug doses.
-
Reduced side effects compared to oral opioids.
-
Programmable delivery for personalized pain management.
-
Improved quality of life, mobility, and sleep.
-
Long-term solution for intractable pain or spasticity.
-
Reversible and adjustable – therapy can be tailored to the patient’s needs.
Risks and Side Effects
Although generally safe, potential risks include:
-
Infection at the surgical site
-
Catheter displacement or blockage
-
Pump malfunction (rare)
-
Overdose or withdrawal if pump is not managed properly
-
Local discomfort at implantation site
With regular monitoring and skilled care, these risks are minimized.
Who is a Candidate?
-
Patients with chronic, severe pain not relieved by conventional treatments
-
Individuals with high opioid tolerance and severe side effects
-
Cancer patients with progressive pain unresponsive to medication
-
Patients with neurological spasticity who do not respond to oral drugs or injections
Effectiveness of Intrathecal Pump Therapy
-
Immediate relief after implantation in most patients.
-
Reduces oral opioid use by up to 90%.
-
Improves daily functioning, mood, and activity level.
-
Allows cancer patients to spend time with family in greater comfort.
-
For spasticity, it reduces muscle stiffness and improves movement.
Intrathecal Pump vs. Other Treatments
| Factor | Intrathecal Pump | Oral Medications | Injections | Surgery |
|---|---|---|---|---|
| Targeted Relief | Direct to spinal fluid | Systemic (whole body) | Local/temporary | Variable |
| Dose Required | Very small | High | Medium | — |
| Side Effects | Minimal | High | Moderate | Moderate–High |
| Duration | Long-term | Short-term | Short to medium | Depends |
Patient Case Studies
-
Case 1: Cancer Pain – A 65-year-old man with advanced lung cancer was unable to tolerate oral opioids due to nausea and drowsiness. After pump implantation, his pain reduced dramatically, allowing him to eat, sleep, and communicate comfortably.
-
Case 2: Spinal Cord Injury – A 42-year-old woman with severe neuropathic pain post-accident regained mobility and reduced her dependence on strong oral drugs.
-
Case 3: Multiple Sclerosis – A 50-year-old man with severe spasticity improved muscle control and was able to resume basic daily activities.
Aftercare and Recovery
-
Hospital stay is usually 1–2 days.
-
Avoid strenuous activities until the surgical site heals.
-
Pump refills are required periodically.
-
Regular follow-ups ensure correct dosage and device functioning.
Future of Intrathecal Pump Therapy
-
Smarter pumps with wireless monitoring.
-
Combination drug delivery for tailored therapy.
-
Integration with AI for automatic dose adjustments.
-
Regenerative medicine to eventually reduce reliance on pumps.
Conclusion
The intrathecal pump implantation is a life-changing therapy for patients suffering from severe, chronic pain or spasticity unresponsive to conventional treatment. By delivering medication directly to the spinal fluid, it provides powerful relief with fewer side effects, restoring dignity, comfort, and mobility.
For many patients, this therapy represents hope—a way to live with less suffering and more freedom, even in the face of complex and chronic conditions.
